Amiculum Blog Article
What defines the ideal therapy today? Insights from PharmaQuest at ISPOR Europe
What defines the ideal therapy today? Insights from PharmaQuest at ISPOR Europe
What defines the ideal therapy today? Insights from PharmaQuest at ISPOR Europe

What defines the ideal therapy today? Insights from PharmaQuest at ISPOR Europe
What do stakeholders really value when it comes to new drug therapies? At ISPOR Europe 2025, we set out to answer this question through PharmaQuest, an interactive survey that challenged respondents to make real-world trade-offs in designing the ‘ideal’ therapy. With strong engagement from ex-payers, key opinion leaders, health technology assessment (HTA) stakeholders and pharma professionals (n=91), the results reveal clear priorities: quality of life (QoL) improvements often outweigh marginal efficacy gains, rare diseases dominate pipeline interest, and there is flexibility on cost-effectiveness thresholds when curative potential is on the table. These insights highlight the trends shaping market access strategies and what this means for the pharmaceutical pipeline.
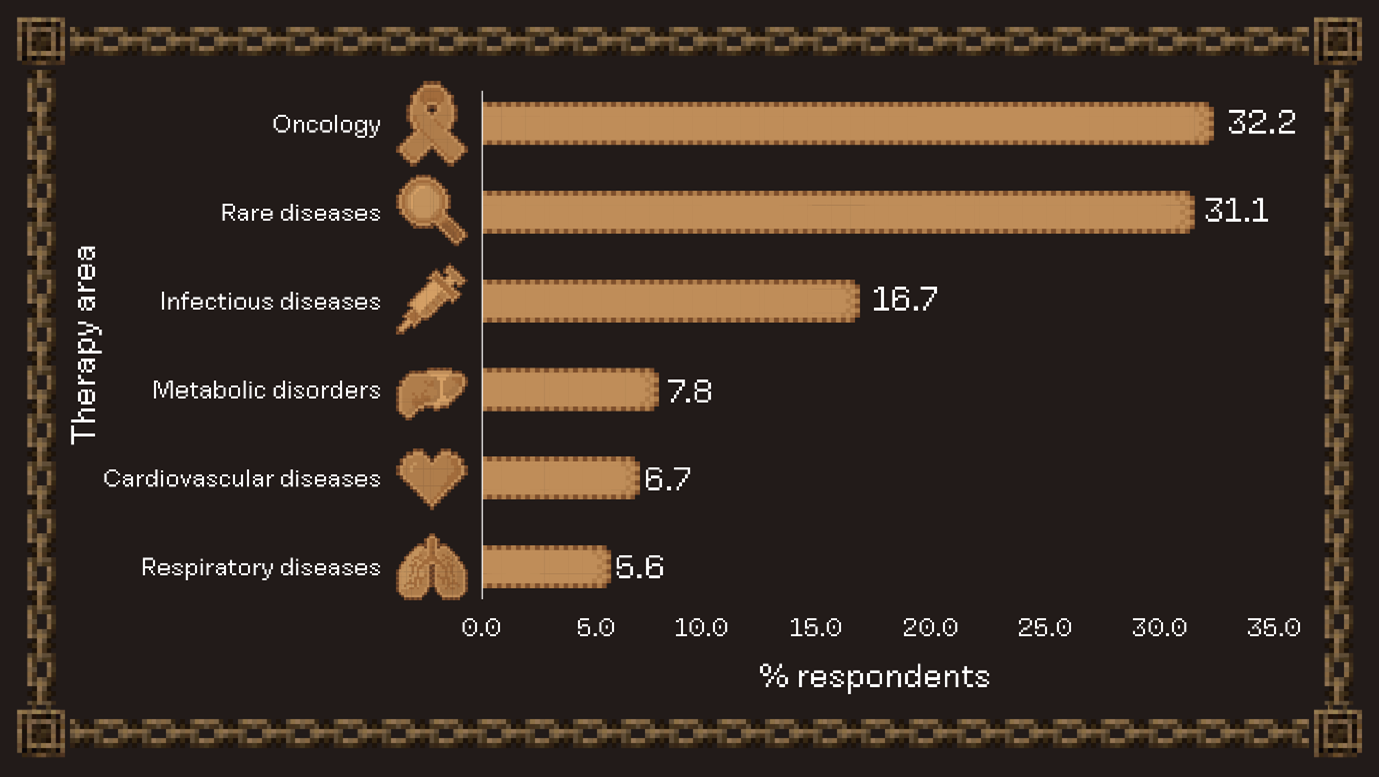
Pipeline priorities: oncology and rare diseases top the list
Oncology was the most popular therapy area selected by respondents, reflecting its position at the forefront of innovation with a robust pipeline. Regulatory incentives, such as the Food and Drug Administration (FDA)’s priority review vouchers, have made rare disease drug development increasingly attractive. Infectious diseases also ranked highly, reflecting ongoing global priorities such as pandemic preparedness and antimicrobial resistance. Metabolic disorders, despite the explosion of interest in glucagon-like peptide-1 agonists, were less popular among respondents, perhaps reflecting a focus on other areas perceived as having greater unmet need or innovation potential.
Summary of responses to question, “Select the therapy area you would prioritize for designing your ideal drug”
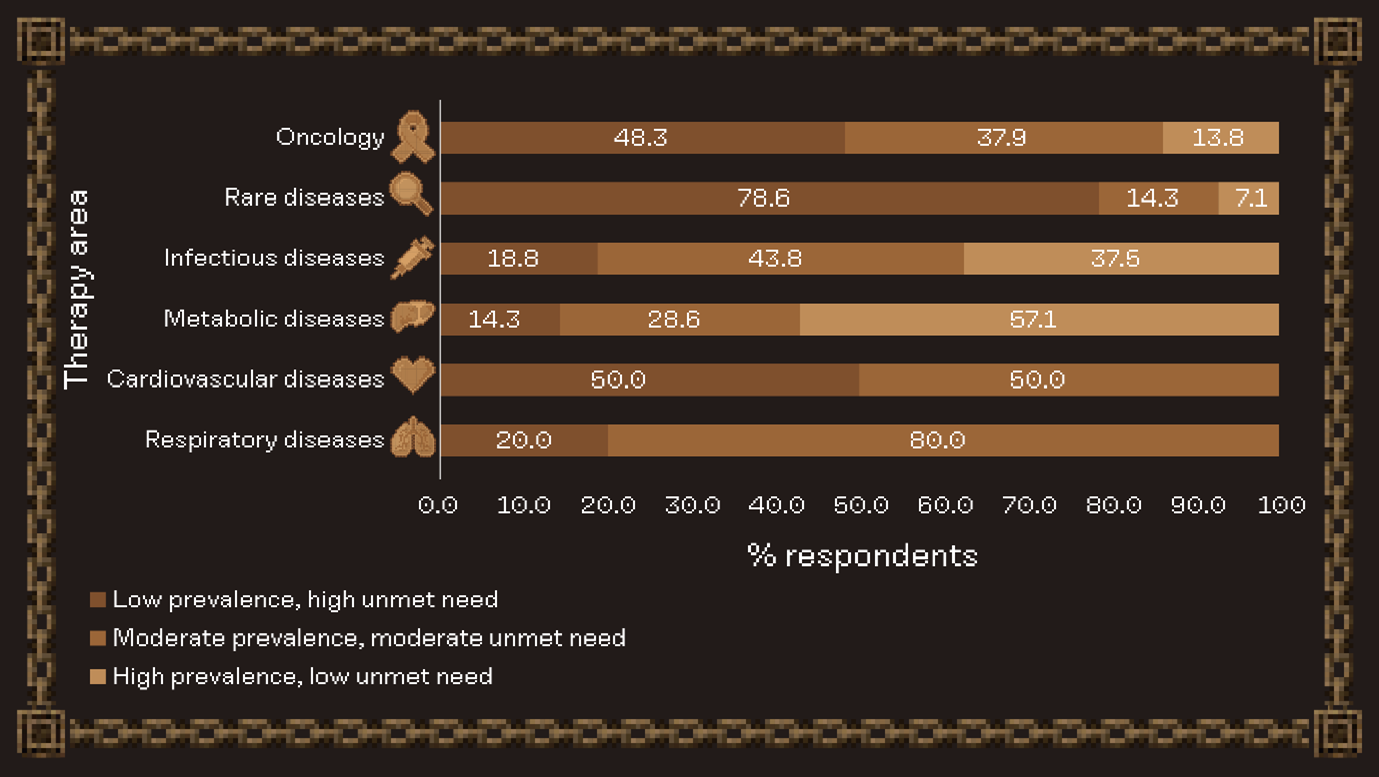
Rare but rewarding: small populations, big opportunities
Our respondents opted for “low prevalence, high unmet need” when asked which area they would prioritize, aligning with rare disease being one of the most popular disease areas. This may also reflect trends in personalized medicine with increasingly small patient populations.
Summary of responses to the question, “Would you prioritize an indication with high prevalence or high unmet need?”
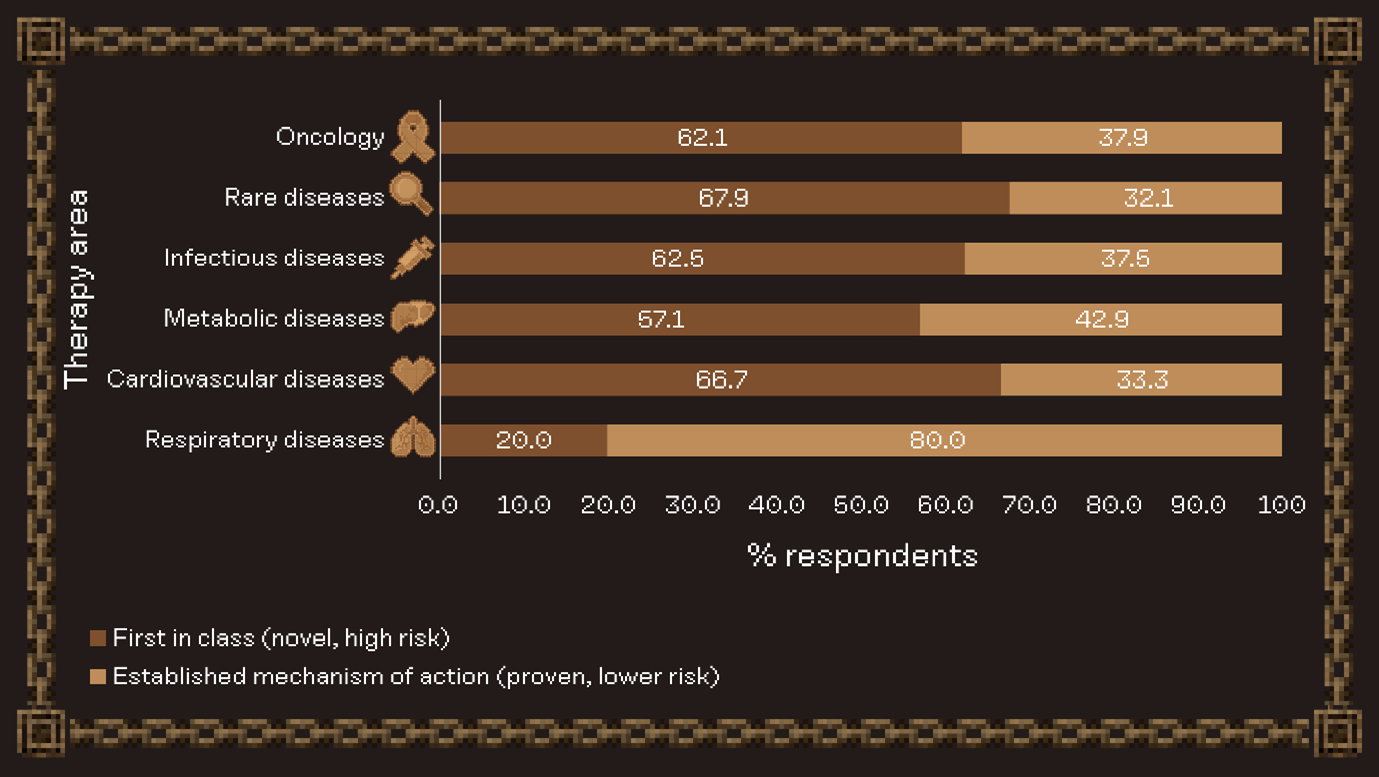
Novelty over certainty: innovation takes the lead
How risk tolerant were our respondents? Across all therapy areas except respiratory disease, preference was for a first-in-class product, suggesting a healthy appetite for risk among respondents. Rare disease had the highest percentage of respondents selecting first in class, reflecting the high unmet need and willingness to accept uncertainty for breakthrough benefit. Conversely, in cost-sensitive, high-volume markets such as respiratory disease, choosing a ‘me-too’ therapy was more popular.
Summary of responses to the question, “Would you prefer a first-in-class product offering higher risk or an established mechanism with lower risk potential?”
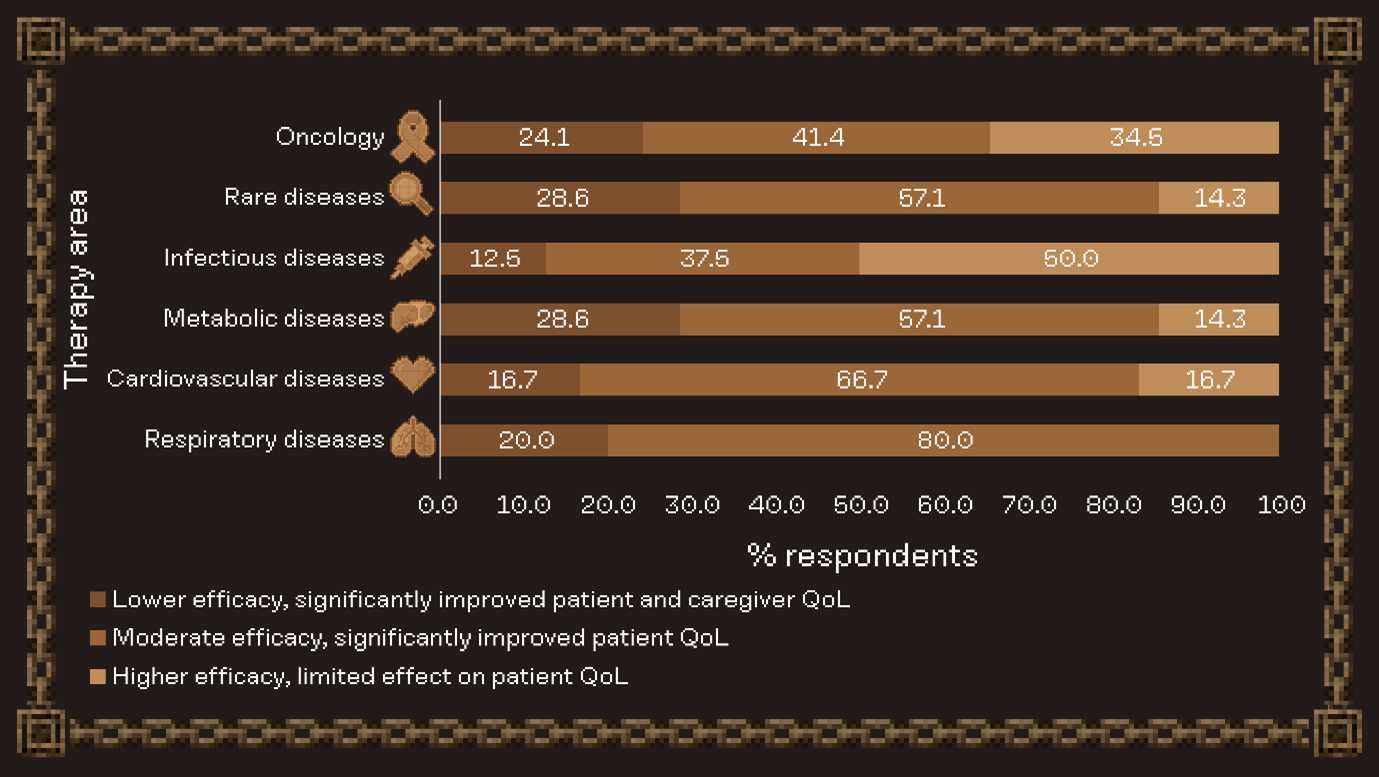
Beyond clinical outcomes: QoL takes priority
Respondents valued therapies that deliver meaningful improvements in QoL, even when efficacy gains were modest. This is especially relevant for cardiovascular and respiratory diseases, reflecting stakeholder focus on holistic outcomes for chronic conditions. Oncology and infectious diseases had the highest percentage of respondents prioritizing higher efficacy, indicating continued emphasis on survival and disease eradication in high-burden therapy areas.
Summary of responses to the question, “Would you prioritize higher efficacy over better QoL (quality of life)?”
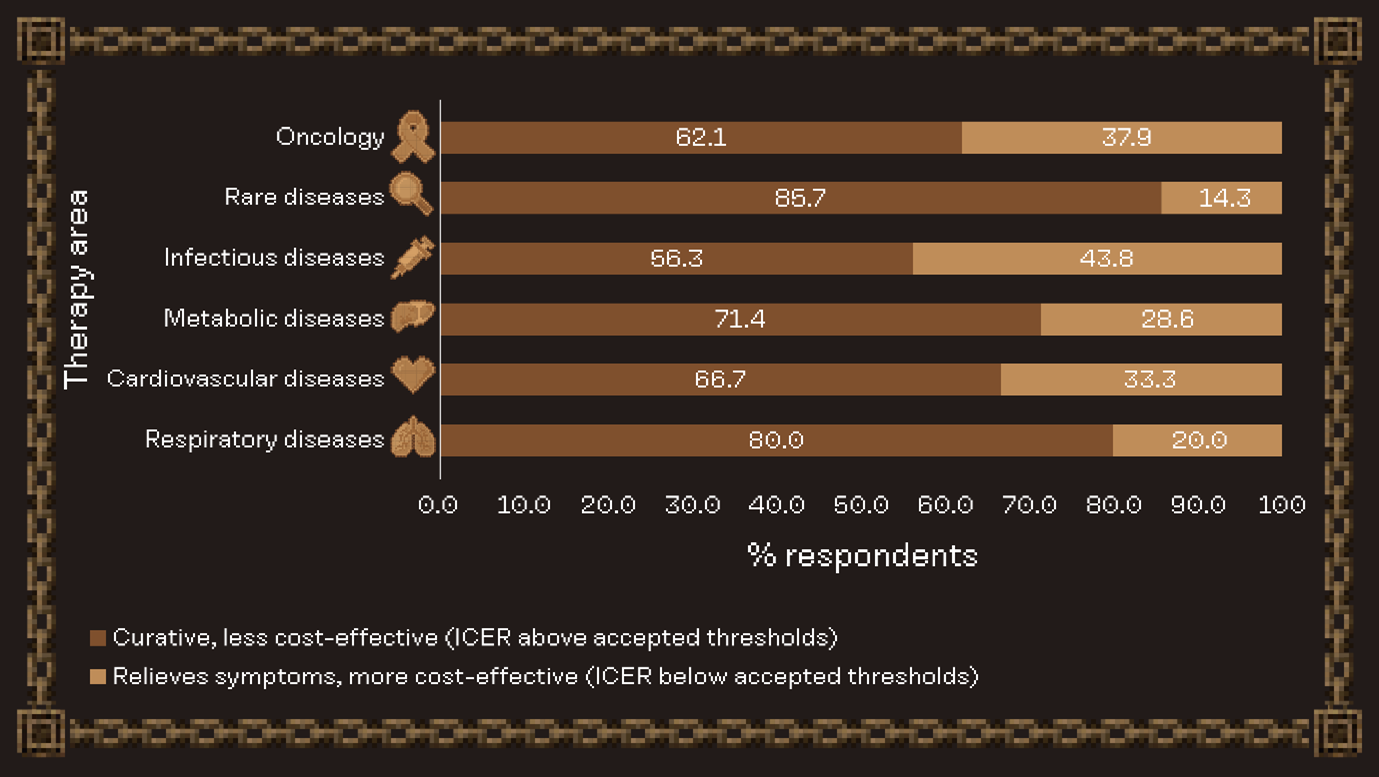
Breaking the incremental cost-effectiveness ratio (ICER) barrier: when cure justifies cost-effectiveness
Stakeholder willingness to accept higher cost-effectiveness thresholds depended heavily on therapeutic context. In rare diseases, respondents prioritized curative treatments even when ICERs exceeded accepted limits, aligning with higher payer flexibility on ICERs when unmet need is high. Conversely, in infectious diseases, cardiovascular diseases and oncology settings, preferences leant towards cost-effective symptom relief, reflecting tighter scrutiny on cost-effectiveness.
Summary of responses to the question, “Would you prefer a product that is curative but exceeds cost-effectiveness thresholds or one that treats symptoms and is significantly more affordable?”
What does this mean for pharma?
These findings reinforce a global trend: HTA bodies and regulators are embedding patient perspectives into evaluation frameworks, as seen in the EU’s Joint Clinical Assessment. Success in market access now hinges on patient-centred evidence, flexible pricing strategies and risk-aligned contracting. Drug developers that anticipate these trends by integrating QoL endpoints, planning for real-world evidence and tailoring value propositions by stakeholder will be best positioned to win in an increasingly competitive landscape.
This research is only a starting point and, while a great snapshot, the sample size was small and the questions used a simple three-choice structure. However, it provides the foundation for future analyses that will explore how these preferences change and differ across geographies and payer archetypes – insights that will inform smarter, more resilient access strategies.
About the author
What defines the ideal therapy today? Insights from PharmaQuest at ISPOR Europe
What defines the ideal therapy today? Insights from PharmaQuest at ISPOR Europe
Contact CTA
Start a conversation
Find out how our experts can address your healthcare communication challenges.
